RETURN & REFUND POLICY:
There will be no refund made on purchases of Lectures-on-Demand or Clinical Videos unless the order was duplicated in error (the customer was charged twice for viewing the same lecture or clinical video). DVDs may not be returned for refund or exchange unless broken upon opening or defective upon viewing.
If the item is still sealed in the original packaging, gIDE will exchange it or offer a refund based on the original method of payment. Products from opened packages will only be exchanged if deemed defective.
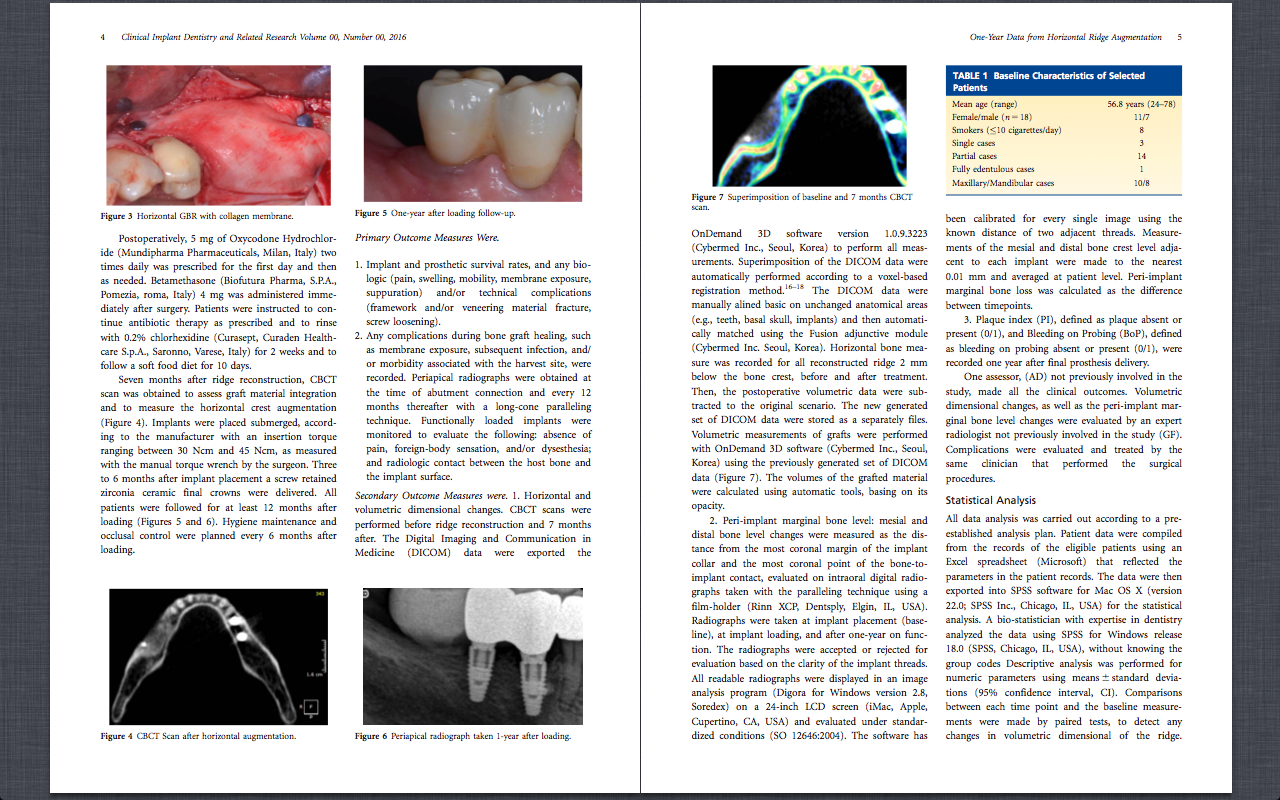
TRAINING CENTER POLICY
The cancellation fee is $750 up to forty-five (45) days prior to each program. After that date, there will be no refunds. As a courtesy, registrants can make a one-time date change within a 12 month period, space permitting.
Registrations received during cyber-monday sales are non-refundable. gIDE may cancel scheduled courses up to thirty (30) days prior to the scheduled start of the course if certain attendance minimums are not met. gIDE is not responsible for reimbursing prepaid (including non-refundable) customer travel costs. It is recommended that customers who elect to incur travel costs for a training course, schedule and purchase airfare and hotel accommodations with this in mind.
Dates and locations are subject to change.
MASTER CLINICIAN PROGRAM CANCELLATION & REFUND POLICY:
Initial deposit is non-refundable.
Initial deposit and 1st installment payment are non-refundable after Session I.
All payments received prior to Session III are non-refundable regardless of participation during sessions III and IV.
In special circumstances, course completion postponement may be approved by gIDE. In such cases, fees may be applied. Refund policy is applied based on date of first enrollment.
Dates and locations are subject to change.
COVID-19 POLICY:
All appropriate measures will be taken to ensure the highest level of safety for faculty, staff, and participants. At all times gIDE will be fully adhering to the regulations of the local government, official health departments, meeting venues, as well as best practices. This will include considerations to class size, the wearing of masks by all, and any other measures that promote safety.
gIDE can elect to cancel or postpone scheduled courses due to the health risks associated with the current pandemic and cannot be held responsible for reimbursing prepaid (including non-refundable) customer travel costs.
Advance Proof Of Vaccination or Negative PCR Test / Antigen Test 24-48 Hrs Before Program Start Date
REPLACEMENT:
In the case of a defective DVD, the product must be returned within seven (7) days and deemed defective by gIDE Customer Support in order to be eligible for a replacement.
DEFECTIVE ITEMS:
If you discover what you believe is a product defect, please contact gIDE Customer Support at +1 310-696-9025. Such a defect, if any, is covered under the terms of your product"s warranty.
Please refer to the warranty information and other supporting documentation that was shipped with your order. (See Product Warranty section below for specific information about gIDE"s product warranties.)
ELECTRONIC SOFTWARE DOWNLOADS:
Using a valid gIDE Account, you can select downloadable software products for purchase in the gIDE catalog. At the time of checkout, we will obtain an authorization on the credit card you have provided.
Once credit card authorization has been received, you will receive instructions in the form of a link to begin viewing your Lecture-on-Demand and/or Clinical Video.
If the transmission is interrupted in any way, you can use your gIDE account and password to begin the process again. Once you complete your order, you will receive a separate email acknowledgment that provides information about your purchase.
If gIDE cannot obtain authorization to charge the credit card submitted, gIDE will send instructions, via email, for completing the purchase. Please read the product information for Electronic Software carefully.
Software purchases are non-refundable. Electronic Software Sales Tax Applicable sales tax will be charged for software purchases where required by local law based on the jurisdiction in which the purchaser resides.
HOLD HARMLESS:
gIDE Institute bears no responsibility for the outcome of patient treatment skills & techniques that are learned from their programs.